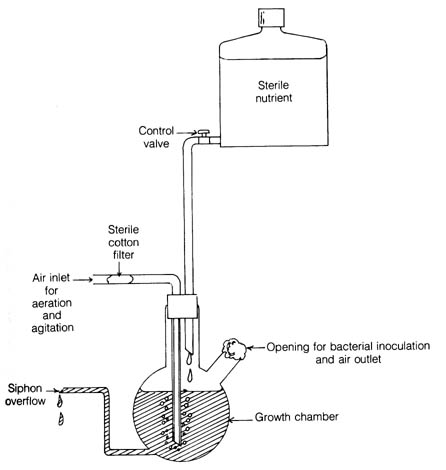
Figure 3.1. Simplified diagram of a chemostat.


3.2 Evaluation of Microevolution (The Special Theory of Evolution)
3.2.1 Empirical Adequacy. The mechanism of the Neo-Darwinian
concepts of microevolution can be documented empirically.

Figure 3.1. Simplified diagram of a chemostat.
a) Mutation as the Raw Material of Evolution. As described
in I.2.6, [177] spontaneous mutations, especially those caused by nucleotide
substitution, may not necessarily be harmful to an organism. They can also
be either neutral or advantageous. The beneficial effects of mutations
have been documented best in microorganisms. For example, certain spontaneous
mutants of Bacillus subtilis, a commonly found soil bacterium, show
an increased growth rate even in the absence of selection. Mutants grew
faster than the normal (wild type) bacteria at 31°C, 34°C, 37°C,
45°C, and 48°C in a defined culture medium that provided only minimum
essential nutrients (1).
Behavior of bacterial mutants like those above is studied in a chemostat (Figure 3.1). The device allows the continuous growth of bacterial culture. The design of the apparatus ensures the constant supply of nutrients [178] and oxygen to the growth chamber as well as the continuous washout of excess bacteria that, if allowed to remain in the chamber, would slow the overall bacterial growth rate. Optimal temperature and oxygen levels for growth are maintained. Theoretically, a pure bacterial culture in the growth chamber could grow for an indefinite period of time if contamination from other microorganisms is avoided. Therefore, the effects of mutation in the pure culture can be followed for many generations. A bacterial strain growing in the chemostat for 10 days at 37°C at a generation time of two hours and at an original density of 5 x 108/ml was replaced by a new bacterial strain that arose from spontaneous mutation. The growth rate of the mutant was five times the rate of the original strain (2).
The cumulative effect of several sequential nucleotide replacement mutations was shown to be harmless or beneficial in the human colon bacterium Escherichia coli. The strain selected contained a mutant locus in the mutator gene (see I.2.6.3.a.3). It was grown in a chemostat for 1400 generations, and during this time a nucleotide replacement rate of about seven transversions per bacterium per generation was calculated. At the end of the growth period approximately 10,000 nucleotide pairs were replaced in the bacterial strains found in the chemostat. It was found that this drastically mutated bacterial strain grew at the same rate as the original bacterial culture in the chemostat (3).
In another experiment that involved the study of competition of a normal strain of bacteria with a bacterial strain mutated in its mutator gene, equal numbers of each strain were introduced into the chemostat at the beginning. The mutant strain was observed to outgrow the wild-type strain 23- to 102-fold in periods ranging from 99 to 777 elapsed generations. Comparable ratios were also observed if initial mutant populations were less than the normal strain. These experiments suggest that the mutant strain that underwent extensive nucleotide substitutions in its own DNA, presumably as a result of a mutated form of DNA polymerase, actually grew faster than the nonmutated strain (4). The cumulative effect of several nucleotide substitutions appeared to be beneficial under the conditions of the experiment.
Irradiation-induced mutations also may be favorable to an organism. A study was made of the effects of x-irradiation on the fruit fly Drosophila birchii. Two identical populations were established from a natural population. One population served as a control while the other population was given 1000 R (Roentgens) of x-ray during each of the first three generations. This dosage of irradiation was enough to induce a large number of mutations. During the initial generations, the number of flies in the irradiated population was smaller than the control population, presumably [179] due to flies dying from harmful mutations caused by radiation. However, in succeeding generations the irradiated populations recovered, and they outgrew the control populations in approximately two years, during which time 30 to 40 generations passed (Figure 3.2, Table 3.1). In the experiment, radiation-induced mutations permit the flies to better exploit the experimental conditions (Table 3.1).
Figure 3.2. Rate of evolution in irradiated and nonirradiated experimental populations of Drosophila birchii. Each of three large populations was divided into two: one was subject to irradiation for three generations, the other was the control. All populations became increasingly adapted to the experimental environment, as reflected in the gradual increase in size. The irradiated populations, however, increased at a faster rate than the controls, evincing that some mutations induced by radiation may be favorable to their carriers. Reprinted, with permission, from Dobzhansky, T. et al. Evolution. San Francisco: W. H. Freeman and Co.; 1977. © 1977 by W. H. Freeman and Co.
b) Selection as the Driving Force for Evolution. The principle of selective medium has been widely used in microbiological laboratories to isolate rare bacterial mutants. When this principle is used, a certain nutrient necessary for the growth of unmutated bacteria is withheld from the selected agar medium; therefore, the only bacteria that grow on this agar plate are strains that are mutated to become independent of the missing nutrient. By the same token, when a drug such as an antibiotic that kills normal unmutated bacteria is added to the agar medium, the only bacteria that survive in the drug-supplemented medium is a strain mutated to acquire drug resistance. By this principle, mutation as rare as one mutant per one billion wild-type bacteria can be detected. When a bacterial culture containing a mutant is introduced into a selective agar medium, the mutant will grow and form a visible colony at the expense of the unmutated bacteria after an appropriate time of incubation. Experimental work with the many bacterial mutants that were originally isolated by using the principle of selective medium has contributed greatly to advances in molecular genetics (1,5).[180] Table 3.1. Rate of evolution of irradiated and nonirradiated populations of Drosophila birchii in the experiment in Figure 3.2.**NOTE: From Proceedings of National Academy of Science, Washington, D.C. 63(3)790-93; 1969.
With the discovery and use of antibiotics to fight bacterial infection, several drug-resistant strains of bacteria have increased in occurrence and are of medical concern. For example, the widespread use of penicillin has fostered an increase in populations of penicillin-resistant Staphylolococcus [181] aureus, a bacterium known to cause numerous infections in humans such as boils and absesses. Higher and higher doses of penicillin must be administered in order to kill the bacteria. These resistant bacteria are a serious problem in hospitals.
The problem that developed after using penicillin is a clear example of a change in the frequency of a gene bringing about drug resistance under strong selective pressure. The drug-resistant bacteria that were derived presumably by spontaneous mutation must have existed at a very low frequency before the widespread use of penicillin. However, penicillin has been so effective in wiping out drug-sensitive bacteria that rare drugresistant mutants in the natural population have increased rapidly. Of course, as demonstrated earlier (I.1.3), the penicillin-resistant mutation is independent of the presence or absence of the drug.
Selection brought about by insecticides is another interesting example of evolutionary change caused by selection. Resistant strains of parasitic insects soon became known after the use of insecticides. For years, the citrus trees in Southern California have been infested by parasitic scale insects. In order to combat the insects, the trees were covered with tents and then fumigated with a cyanide poisonhydrocyanic acid. Later it was observed that cyanide-resistant varieties of scale insects had replaced the original cyanide-sensitive varieties.
The development of DDT resistance in household pests was also caused by selection after widespread use of the insecticide DDT. When DDT first came into general use in 1945, it was very effective in controlling the proliferation of houseflies, but soon resistant varieties of flies appeared. Under the strong selective pressure of DDT, resistant strains of flies quickly became well established and completely replaced sensitive strains in certain localities.
One of the best-established cases demonstrating the effect of natural
selection in changing the gene frequency of a population is industrial
melanism in the peppered moth Biston betularia, which flies
by night and rests on tree trunks by day. Since the middle of the nineteenth
century, the numbers of light and dark pigmented varieties of B. betularia
have
been compared in rural and industrial regions of England. In the latter
areas the vegetation had been blackened by pollution, and the frequency
of light-colored moths was very low; however, the frequency of the light
varieties was high in the unpolluted coral areas. In some heavily polluted
localities the dark varieties almost totally replaced the light varieties.
A similar phenomenon was observed in 100 other species of moths in England.
Professor H. B. D. Kettlewell demonstrated that the basis of the [182] change in frequency of the gene controlling the moth's pigmentation was their need for protective coloration against their natural predators such as birds (6). In rural areas he noticed that dark pigmented moths resting on tree trunks with light colored lichens (a fungus and alga growing together) were conspicuous to birds while the light varieties were camouflaged. In the industrialized areas lichens were scarce, having been killed by pollutants. In these regions the dark varieties of the peppered moth matched the color of the polluted tree trunks, making it difficult for predators to detect them. This phenomenon was very striking in the industrialized city of Birmingham. In contrast, the presence of any light colored moth resting on a tree trunk in the Birmingham area was obvious (Figure 3.3).Figure 3.3. Left: The peppered moth, Biston betularia, and its melanic form, carbonaria, at rest on a soot-covered oak trunk near the industrial city of Birmingham, England. The carbonaria form is much less conspicuous than the typical peppered form, which is very conspicuous. Right: The same forms resting on a lichened tree trunk in unpolluted countryside. The typical form is much less conspicuous than the melanic. Reprinted, with permission, from Kettlewell, H. B. D., Department of Zoology, South Parks Road, Oxford, OX1 3PS. Arrows point to the moths.
Kettlewell theorized that birds selectively pick out the conspicuous varieties in both the polluted and nonpolluted environments, thereby contributing to the change in the frequency of the genes, controlling pigmentation. In order to test this hypothesis, he raised thousands of light- and dark-pigmented peppered moths, marked each with a dot of [183] paint, and released them into the polluted (industrialized) and nonpolluted (nonindustrialized) areas of England. He then set out to recapture these moths. In the nonindustrialized areas, he managed to recapture 14.6% of the light varieties but only 4.7% of the dark varieties. In the industrialized areas, he was able to capture only 13% of the light varieties; however, 27.5% of the dark varieties were recovered.
In another series of experiments Kettlewell was able to photograph birds preying on moths that he placed on tree trunks. He demonstrated unequivocally that birds eat more light varieties in the polluted areas and more dark varieties in the nonpolluted areas.
Direct experimental tests, as well as the discovery of dark varieties in regions far away from the industrialized areas, suggested that the dark varieties did not arise from mutations induced by particulate matter, such as heavy metals, emitted from industrial plants. Industrialization apparently changed the environment, resulting in natural selection favoring survival of dark varieties and loss of light varieties. Thus the natural selective force of predation in a changing environment caused by industrialization was responsible for microevolution in moths.
c) Balancing Selection. Natural selection is not only capable of inducing changes in gene frequencies, it can also maintain the frequency of a harmful gene in a population, provided that this gene is advantageous for its carriers. This phenomenon is called balancing selection. If two or more alleles are maintained at a single genetic locus, the phenomenon is called balanced polymorphism.
An excellent example of balanced polymorphism is the distribution of
the gene responsible for sickle cell anemia. As already discussed
in I.2.6.2, the substitution of a nucleotide in the gene encoding for the![]() chain is called hemoglobin molecule changes the configuration of the protein
reducing its solubility and causing the stacking of adjacent molecules.
Hemoglobin containing this aberrant
chain is called hemoglobin molecule changes the configuration of the protein
reducing its solubility and causing the stacking of adjacent molecules.
Hemoglobin containing this aberrant ![]() chain is called hemoglobin S (HbS) and is distinguished from the
normal hemoglobin
A (HbA).
chain is called hemoglobin S (HbS) and is distinguished from the
normal hemoglobin
A (HbA).
Sickle cell anemia patients are homozygous for the S gene (SS). Individuals heterozygous (AS) for the S gene are called carriers, having no apparent symptoms of the disease under normal levels of atmospheric oxygen. Therefore, the S gene is recessive and is lethal only when it is expressed in the homozygous (SS) state. Normally, if a gene is harmful, it will be selected against, and its frequency will diminish. However, in large areas of the world, the gene frequencies of S are much higher than would be expected on the basis of the selection against a lethal trait. This can be seen in Figures 3.4 and 3.5.
After much research it became apparent that the distribution of the S [184] gene has strong correlation with the occurrence of malaria. The reason behind this correlation is that individuals heterozygous for the S gene (AS genotype) are resistant to malaria. When the malaria parasite in the red blood cells consumes all the oxygen carried by the cell, the AS cells become sickle shaped. (Red blood cells in normal individuals never sickle except at high altitudes where oxygen is limited.) The malaria parasite becomes trapped in the sickle cells, which in turn attract phagocytic cells of the defense system of the body that destroy both the cells and parasites. Thus, the AS genotypes makes a carrier resistant to malaria.
Since the heterozygote of a harmful gene becomes advantageous to its
carrier, this phenomenon is also called positive heterosis (overdominance).
In
Africa where malaria is prevalent, the frequencies of genes A and
S
are in equilibrium. This is achieved by balancing the disadvantage of the
A
gene with the disadvantage of the S gene through comparable selection
pressure against each trait. If the gene frequency of S falls, due
to the early death of SS individuals who fail to reproduce, the
number of [185] AA individuals (normal) increases, and they are
highly susceptible to malaria. Malarial deaths of AA individuals
lowers the frequency of the A gene in the next generation. The lowered
A
gene frequency means a rise in the frequency of the
S gene resulting
in more SS individuals who are likely to die of sickle cell anemia.
Thus, the sequential increase and decrease in frequency of both genes results
in a stable frequency over many generations in a malaria environment.
Figure 3.4. Distribution of the sickle-cell gene in Africa, the Middle East, and India. Sickle-cell gene is commonest in populations of tropical Africa; in Zaire, for example, the S gene frequency is about 18%, which means that some 30% of the population carry the AS trait. The sickle-cell gene is also found in the Mediterranean, particularly in Greece and Turkey, and in northwestern Africa, southern Arabia, Pakistan, India, and Bangladesh. Individuals who carry the AS trait are more resistant to malaria than others. Reprinted, with permission, from Cavalli-Sforza, L. L. The genetics of human populations. Sc. Am. (Sept.) 83; 1974.
Figure 3.5. Distribution of malignant malarias caused by the parasite Plasmodium falciparium. Malaria was common in the 1920s in the parts of the Old World indicated on this map. Overlap with sickle-cell gene distribution is extensive. In many regions where malaria is prevalent but HbS is not, other mutant hemoglobins are commonly found. Reprinted, with permission, from Kirk, D. Biology today. 2nd ed. New York: Random House, 1975.
In America the A and S gene are not in equilibrium.
The presence of an S gene is disadvantageous because malaria has
been eradicated due to aggressive public health programs, and the frequency
of the A gene continues to increase as the environment selects against
the S gene. This is directive selection (see I.1.4.5.).
3.2.2 Rational Coherency.
a) Domestication. Domestication of animals and plants has been
practiced from early human civilization, and breeders have been able to
produce altered and improved breeds of both domestic animals and cultivated
[186] plants. In the 1800s Darwin became interested in these alterations.
He
Figure 3.6. Variation among the breeds of domestic pigeon, a subject of investigation by Darwin that confirmed for him the strong influence on variation that can be exerted by natural selection. Reprinted, with permission, from Stebbins. The pigeon. Levi Publishing Co., Inc. (n.d.).
noted the changes in the anatomical features in many domesticated
pigeons as compared with wild pigeons. He found that some features of domestic
breeds differed from each other to a greater degree than differences between
species or even families of wild birds (Figure 3.6). Although the process
of change of these features that must have occurred during the history
of domestication cannot be observed, it is reasonable to assume that the
artificial selection pressure imposed on the breeds by the breeders contributed
to changes in anatomical features. It is also possible that changes were
brought about by inbreeding depression (the selection of homozygous
genotypes when heterozygotes are mated with each other in an inbreeding
population) that could lead to reduction in the diversity of the breeds.
Nevertheless, the breeders' experience at least provides a [187] rational
basis for the theory that selection can bring about changes in gene frequencies.
b) Speciation in Drosophila. The formation of new species (speciation) cannot be observed in the laboratory or in nature, because of its presumed slow pace. Yet the fact that speciation is occurring or has occurred is a reasonable inference that can be drawn from experimental models and natural populations. The four stages of speciation are believed to be: (1) division of a single population into two identical populations isolated from each other by a physical barrier, (2) the formation of a reproductive barrier between two populations, (3) the encounter of the two reproductively isolated populations, and (4) the independent evolutionary development of each population.
The evidence for the above stages of speciation is sparse, especially for the first stage. However, a rare case of reproductive barrier formation in natural populations of the fruit fly Drosophila was reported by Prakash (7). He collected flies of Drosophila pseudoobscura in Bogota, a city located in the highland of Colombia, South America. When the females from the Bogota population were crossed with males from any other location, the males in the first filial generation (F1) were completely sterile. The reciprocal cross produced normal male offspring. These results are similar to that of the cross between two species of DrosophilaD. pseudoobscura and D. persimilis. The cross produces F1 mates with very small testes when the cross is made in one direction but not when made in the other direction. Thus the sterility of the male offspring produced by the females in the Bogota population with the males from other populations may be the first stage of speciation.
The above interpretation is strengthened by the fact that D. pseudoobscura was believed to be scarce in Colombia before 1960 after an intensive search located very few examples. However, they were found to gradually appear and increase in frequency from 1960 to 1962. The populations used for Prakash's studies were collected in late 1967. Thus D. pseudoobscura may have been introduced into Bogota from North or Central America before 1960 where they later proliferated and populated the Colombian highland. However, it is possible that population numbers of D. pseudoobscura were at a low level in Colombia for a long time and thus went undetected. The evidence strongly suggests that D. pseudoobscura in Bogota was undergoing speciation due to the isolation from the main region of its distribution. Furthermore, the process of speciation seemed to be accompanied by minimal genetic differentiation. Thus, this example provides support for the view that isolation and selection contribute to the evolution of a new species.[188]
c) Distribution of Skin Color. The distribution of skin pigments among different human populations is also an important demonstration of the effects of natural selection on phenotypic traits (8). The distribution around the world of human skin color is shown in a very generalized way in Figure 3.7. The map is plotted in terms of skin color based on minimal exposure to the sun.
The adaptive values of each skin color is readily observable. The melanin particles, which are responsible for dark pigmentation, are distributed throughout the outer layers of the skin to protect the deeper layers from damage by ultraviolet irradiation from the sun. Dark-skinned people are native to the tropics and have thicker skin that contains more melanin than light-skinned Europeans.
In the United States, the frequency of skin cancer is seven to eight times greater in whites than in blacks living in the same urban areas in various latitudes. Although white skin tends to reflect more sunlight, the thicker dark skin is more resistant to damage by sunburn. Furthermore, the fact that dark skin absorbs heat faster means that the individual having it sweats more. The turning on of the evaporative cooling mechanism may also be an evolutionary advantage. Although the complete evolutionary significance of skin pigmentation is not known, the distribution of skin color as shown in Figure 3.7 is suggestive of its adaptive values.
The two darkest categories of skin color are concentrated around the equator in the Old World. This is to be expected because dark skin color protects against intense tropical solar radiation. People of Caucasoid origin living near the tropical zones differ from their Caucasoid relatives in the temperate zones by having much darker skin. Mongoloids living in temperate zones, presumably the place of their origin, have light brown skin that contrasts with the much darker skin of equatorial Mongoloids.
In areas below the tropics of Africa where the dense rain forests have top canopies as high as 200 feet and multilayered stories of foliage, the amount of sunlight reaching the ground is greatly reduced. Living in perpetual shade, the Negroid people in these areas show yellowish to reddish brown skin color, with much less melanin than is found in other Negroid races. [189]
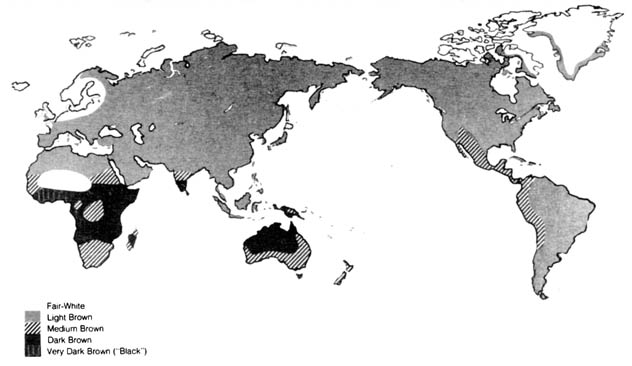
Figure 3.7. Unexposed skin color. Reprinted, with permission, from Birdsell, J. B. Human evolution. Chicago: Rand McNally; 1975. Reprinted, with permission, from Professor Birdsell. Department of Anthropology, UCLA.
People with the least skin pigmentation are found in extreme northwestern
Europe. This region is believed to have been covered by a glacier approximately
12,000 to 15,000 years ago. The inhabitants must have migrated to this
area after the retreat of the glacier. Today the area has an oceanic climate
characterized by cloudy and rainy summers with little intense sunlight.
It has been theorized that the light skin of the northwestern Europeans
is an adaptation that allows maximum absorption of [190] sunlight needed
for the synthesis of vitamin D, a necessary compound that allows the body
to absorb dietary calcium. Natural selection may have favored a reduction
of melanin in these people to a point that almost amounts to partial albinism.
The above hypothesis is highly plausible in view of the correlation of skin color with the weather. The skin color of the vast majority of humanity is light brown, presumably due to the fact that they live outside of the tropics. There is evidence to show that the light brown skin pigmentation does adequately protect these people against solar radiation in the areas where they live.
Before the recent immigration from the Old World, natives of North and South America were intermediate in skin color. Sometimes they had a reddish or ruddy component, hence the term red man (commonly used for Indians). It is believed that native Americans are descendants of a certain Mongoloid race who migrated to North America through the Bering Strait more than 10,000 years ago. The Indian populations also show some selective gradients of pigmentation despite their recent arrival in the Americas. The darkest pigmentation seems to have developed in the Indian population living along the Colorado River where it flows through the hot desert valley on its way to the Gulf of Mexico. Other dark varieties of the Indian race can be found in tropical areas of South America, except for the dense shady forest environment of the Amazon Basin. In contrast the Eskimos of the Arctic and Alaska are lighter in skin tone, experiencing the least exposure to solar radiation.
It is sufficient to conclude that the selective force of solar radiation for protective skin color apparently has produced the spectrum of skin pigmentation in the Old World. However, the relatively recent habitation of the New World has not allowed sufficient time for selection to produce a total regional adaptation.
In summary, microevolution is supported by evidence from studies on the positive effects of bacterial mutation, selection of traits in artificial breeding of domestic animals, fertility changes in insect populations, directive selection against sickle cell anemia in U.S. blacks, and adaptation of selected skin color patterns to specific environmental areas. These examples are both empirically adequate and rationally coherent.
References 3.2
1. Pun, P. P. T.; Murray, C. D.; Strauss, N. J. Bacteriol. 123:346; 1975.